In the field of organoid culture, breaking the bottlenecks of cell proliferation and differentiation efficiency and optimizing technical workflows for staining and detection have long been key research priorities for scientists. The BP4000 Spheroid Generation Platform from Aorui Technology has pioneered a new approach to human colorectal cancer organoid culture through cutting-edge technological innovation, and its technical value has been validated by extensive practical applications.
Below is the latest practical case of human colorectal cancer organoid construction based on the BP4000 Platform:
1. Construction of Human Colorectal Cancer Organoids
1. Collect human tumor tissue fragments and place them in 50 mL centrifuge tubes containing 45 mL of ice-precooled adDMEM/F12 (supplemented with 100 U/mL penicillin-streptomycin, 10 mM HEPES, 1×GlutaMAX) per tube. It is recommended to add 10 μM ROCK inhibitor (Y-27632) to the adDMEM/F12 medium to enhance cell viability. Store tissue samples in this medium at 4 °C until dissociation.
2. Under a dissecting microscope, remove non-epithelial components as thoroughly as possible using surgical scissors or a scalpel and forceps.
3. In a 10 cm cell culture dish, cut the tissue into 1-3 mm³ fragments with surgical scissors or a scalpel, then transfer the fragments to a 15 mL centrifuge tube.
4. Prepare tissue digestion solution according to Table 1 (fresh preparation for immediate use) and prewarm it at 37 °C. Add the prewarmed digestion solution to the centrifuge tube with tissue fragments and incubate on a shaker at 37 °C.
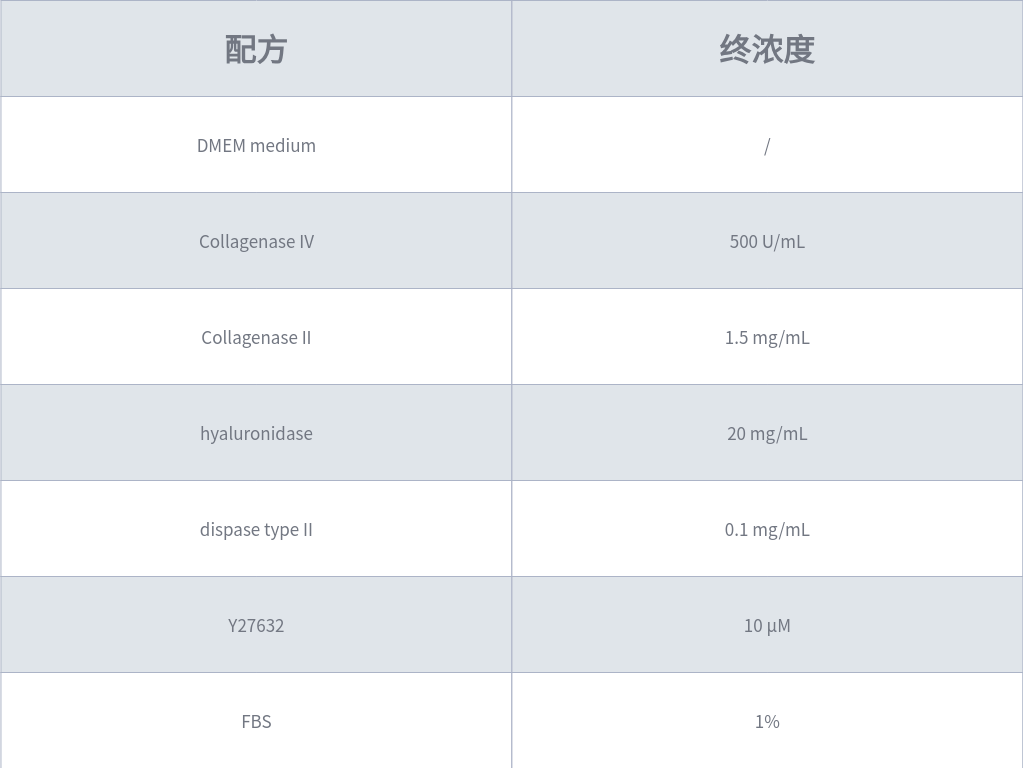
Table 1: Formulation Example of Colorectal Tissue Digestion Solution
5. Once the mixture becomes turbid and residual tissue fragments are disrupted, pipette up and down 10-20 times to further facilitate tissue dissociation. Add 10 mL of adDMEM/F12 (supplemented with 100 U/mL penicillin-streptomycin, 10 mM HEPES, 1×GlutaMAX) to the top of the mixture, then centrifuge at 200 × g for 5 minutes at 4 °C to wash the cells.
6. Resuspend the cell pellet in 10 mL of adDMEM/F12 (supplemented with 100 U/mL penicillin-streptomycin, 10 mM HEPES, 1×GlutaMAX) and filter through a 70-100 μm cell strainer.
7. Centrifuge the resuspended and filtered cells at 200 × g for 5 minutes at 4 °C. Aspirate the supernatant and resuspend the pellet in complete organoid medium containing 70% Matrigel (Corning #356231). Drop 50 μL of the cell-Matrigel mixture into each well of a 24-well plate using a pipette.
8. Incubate the 24-well plate with the seeded organoid mixture in a 37 °C incubator for 10 minutes to allow Matrigel solidification.
9. Gently add 500 μL of complete organoid medium to each well and culture the plate in a humidified incubator at 37 °C with 5% CO₂.
10. Change the medium every 2-3 days by carefully aspirating the old medium from each well and adding fresh prewarmed medium. Mature colorectal cancer organoids can be observed after 5-7 days of culture.
2. Preparation Before Bioprinting
1. Pre-coat the wells of the target plate (for bioprinting) with 70% Matrigel (diluted in adDMEM/F12) according to the volumes recommended in Table 1. Add sterile water to the blank surrounding wells to maintain humidity across the entire plate. Incubate the plate in a 37 °C incubator for 45-60 minutes to solidify the Matrigel. Turn on the humidifier 30 minutes in advance to maintain the humidity in the biosafety cabinet at 70-80%.
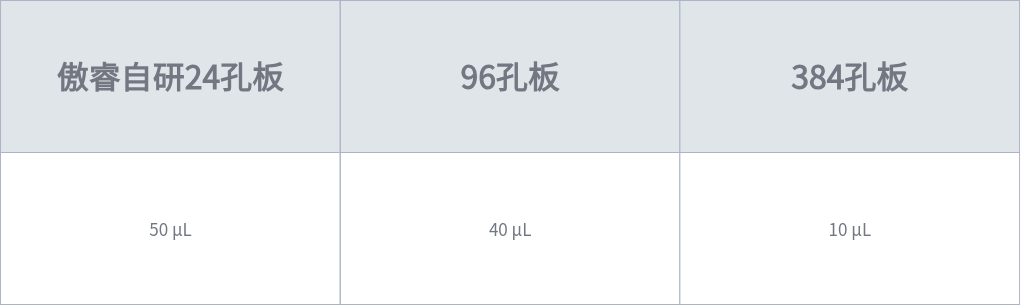
Table 1: Volume of Matrigel for Pre-coating in Different Plates
2. Ensure the humidity in the biosafety cabinet is maintained at 70-80% with the humidifier running for 30 minutes prior to operation.
3. Preparation of Human Colorectal Cancer Organoid Single-Cell Suspension
1. For well-cultured human colorectal cancer organoids in a 24-well plate, carefully aspirate the medium without disrupting the Matrigel dome. Add 400 μL of TrypLE Express to each well and incubate at room temperature (15–25 °C) for 1 minute.
2. Pre-wet a 1 mL pipette tip with TrypLE Express and pipette up and down 20 times to disrupt the solidified Matrigel dome.
3. Incubate the plate in a 37 °C incubator for 5-10 minutes.
4. Using a 1 mL pipette tip pre-wet with TrypLE Express, transfer the culture to a 15 mL centrifuge tube.
5. Centrifuge at 290 × g for 5 minutes at 2–8 °C and carefully remove the supernatant.
6. Resuspend the cell pellet in 10 mL of cold adDMEM/F12 (2–8 °C), then centrifuge at 200 × g for 5 minutes at 2–8 °C. Gently aspirate the adDMEM/F12 supernatant without disturbing the cell pellet.
7. Add the same volume of TrypLE Express as used in Step 2 to resuspend the cell pellet, and incubate in a 37 °C incubator for 5-10 minutes until most organoids are dissociated into a single-cell suspension.
8. Centrifuge at 290 × g for 5 minutes at 2–8 °C and carefully remove the supernatant.
9. Resuspend the cell pellet in 10 mL of cold adDMEM/F12 (2–8 °C), then centrifuge at 200 × g for 5 minutes at 2–8 °C. Gently aspirate the adDMEM/F12 supernatant without disturbing the cell pellet.
10. Resuspend the final cell pellet in adDMEM/F12 and adjust the cell density to 1×10⁶ cells/mL.
4. Bioprinting and Seeding of Human Colorectal Cancer Organoids
1. Turn on the bioprinter and press the homing button to reset the device.
2. Install the print head and perform print head priming according to the operation manual.
3. Place the Matrigel-precoated plate (96-well plate in this example) on the plate holder of the bioprinter.
4. Aspirate the sterile PBS from the print head sample reservoir, then draw 30 μL of the prepared cell suspension and slowly add it to the reservoir for organoid bioprinting, with 2 printing cycles per well (approximately 500 cells). For the control group, the traditional Matrigel drop method was used, with 5 μL Matrigel drops per well (approximately 500 cells).
5. Incubate the bioprinted plate in a 37 °C incubator for 1 hour.
6. Slowly add complete organoid medium to the plate and place it in a 37 °C incubator for continuous culture. The culture results are shown in Figure 1.

5. Immunofluorescence Staining of Organoids
This protocol is optimized for organoids cultured after bioprinting-based seeding.
1. Remove the medium from the wells and gently add 200 μL of calcium- and magnesium-free PBS for washing, repeat twice to avoid damaging the organoid structure.
2. Aspirate the PBS and add 200 μL of 4% paraformaldehyde (PFA), incubate at 4 °C for 45 minutes.
3. Aspirate the PFA and gently add 200 μL of calcium- and magnesium-free PBS for washing, repeat three times.
4. Aspirate the PBS and add 200 μL of OWB (formulation: 1 mL Triton X-100, 2 g BSA, diluted to 1 L with PBS), block at 4 °C for 30 minutes.
5. Aspirate the OWB and add 100 μL of primary antibody diluted in OWB, incubate overnight at 4 °C.
6. On the following day, aspirate the primary antibody-OWB solution and add 200 μL of OWB for washing, repeat three times.
7. Aspirate the OWB and add 100 μL of secondary antibody diluted in OWB, incubate at room temperature for 1.5 hours in the dark.
8. Aspirate the secondary antibody-OWB solution and add 200 μL of OWB for washing, repeat three times.
9. Aspirate the OWB and add OWB containing 1×DAPI, incubate at room temperature for 15 minutes in the dark.
10. Aspirate the DAPI-OWB solution and add 200 μL of OWB for washing, repeat twice.
11. Image the stained organoids under a microscope. Figure 2 shows the Ki67 staining results of bioprinted human colorectal cancer organoids captured by a high-content imaging system (PE).
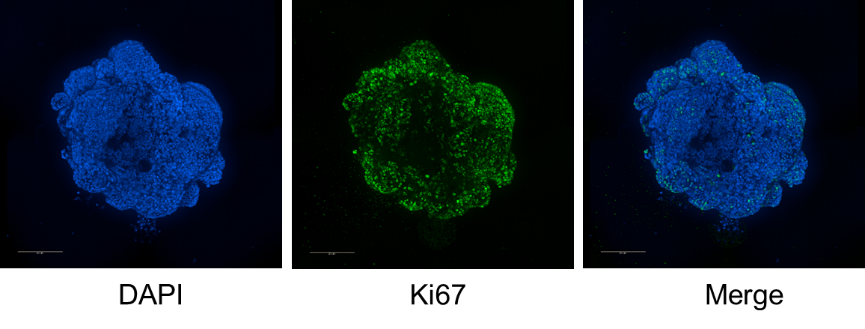
Figure 2: Ki67 Staining of Bioprinted Human Colorectal Cancer Organoids at 14 Days of Culture (Scale bar = 200 μm)
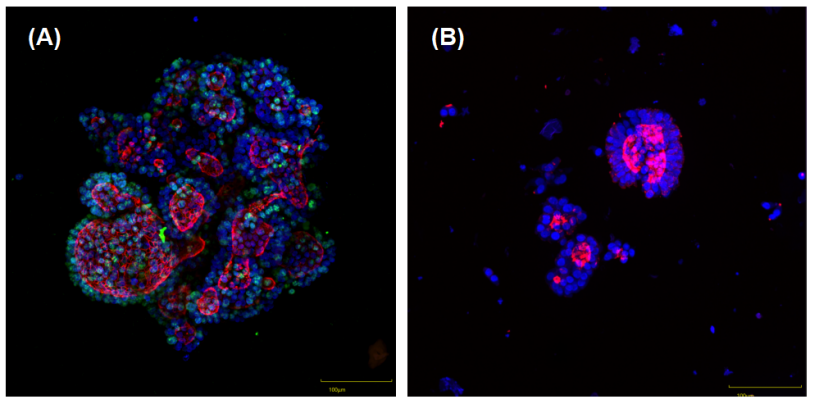
Figure 3: Immunofluorescence Staining of Human Colorectal Cancer Organoids (Green: Ki67, Red: F-actin, Blue: Cell Nucleus)
(A) Bioprinted organoids: Fixed and stained directly, with intact overall morphology, clear staining background and simplified operation.
(B) Matrigel drop-derived organoids: Fixed and stained after Matrigel digestion, with frequent organoid loss or fragmentation during the digestion process.
Product Overview: BP4000 3D Spheroid Generation Platform

3D细胞球构建平台 BP4000
Independently developed by Aure Technology, the BP4000 3D Spheroid Generation Platform integrates droplet control and precision bioprinting technologies to provide a standardized solution for organoid culture. Designed with the core concepts of aggregation bioprinting and digestion-free staining and detection, the platform enables quantitative seeding of single cells or cell clusters on the surface of extracellular matrix (ECM) such as Matrigel, and constructs 3D cell arrays with uniform particle size and controllable spatial distribution through automated procedures. Technically, its core modules include a high-precision droplet generation system, an intelligent temperature-controlled culture unit, and a robotic arm platform compatible with multi-specification plates, meeting the full-process needs from basic research to high-throughput drug screening.
01
Advantage 1: Breaking the Bottleneck of Cell Proliferation and Differentiation Efficiency
In traditional organoid culture, uneven cell distribution and insufficient local cell density are the key factors restricting proliferation and differentiation efficiency.
The BP4000 Platform adopts an advanced aggregation bioprinting mode: through precise droplet control technology, cells are seeded on the Matrigel surface at an optimized local density to form standardized cell arrays. In the human colorectal cancer organoid culture experiment, the aggregation mode of 2 printing cycles per well (≈500 cells) on the BP4000 Platform significantly improved the spatial aggregation of cells compared with the traditional Matrigel drop method.
This high-density aggregation bioprinting has achieved a remarkable improvement in growth efficiency. Experimental data show that colorectal cancer organoids cultured on the BP4000 Platform exhibit a dramatic increase in proliferation and differentiation rate compared with the traditional method. As clearly shown in Figure 1, bioprinted organoids form mature structures within 5-7 days of culture, while organoids from the Matrigel drop method not only have a longer growth cycle but also show significantly inferior structural integrity and consistency.
The cellular microenvironment formed by aggregation bioprinting effectively promotes intercellular signal transduction and material exchange, providing an ideal condition for the rapid growth and functional differentiation of organoids. This efficient culture mode wins valuable time for subsequent drug screening and mechanism research.
02
Advantage 2: Optimizing Staining and Detection Workflows
Immunofluorescence staining is an important technique for analyzing cell function and molecular expression in organoid research. For organoids cultured by the traditional Matrigel drop method, Matrigel dissolution is required before staining — this process often causes organoid loss or damage, seriously affecting the accuracy and reliability of detection results.
The BP4000 technology has completely innovated this workflow: bioprinted organoids can be directly subjected to immunofluorescence staining without Matrigel dissolution, bringing multiple advantages:
Preservation of organoid structure: Avoids damage to the organoid structure during Matrigel dissolution. As intuitively shown in Figures 2 and 3, bioprinted organoids maintain intact overall morphology, while the Matrigel drop method is prone to organoid loss or fragmentation after Matrigel digestion.
Simplified operation and reduced cost: Direct staining eliminates redundant experimental steps, reducing both operation time and labor costs.
High-quality staining results: This method yields stronger staining signals and a clearer background. As shown in Figure 2, the Ki67 staining of bioprinted human colorectal cancer organoids at 14 days of culture shows clear labeling of cell nuclei, Ki67 and F-actin, providing high-quality samples for accurate quantitative analysis.
The digestion-free staining technology of the BP4000 Platform not only improves experimental efficiency but also ensures the authenticity and reliability of detection results, pioneering a new path for organoid functional research.
03
Advantage 3: Enabling More Efficient and Precise Organoid Research
From improving proliferation and differentiation efficiency via aggregation bioprinting to optimizing detection workflows through digestion-free staining, the technical advantages of the BP4000 Spheroid Generation Platform in human colorectal cancer organoid culture not only solve the pain points of traditional methods but also drive organoid research toward higher efficiency and precision.
Its standardized and automated operation mode provides strong support for the construction of large-scale organoid biobanks and high-throughput drug screening, and has broad application prospects in the fields of tumor precision medicine and novel drug research and development. With continuous technological innovation and improvement, the BP4000 Platform is expected to play an important role in the culture and research of more types of organoids, bringing more breakthroughs to life science and medical research.

We specialize in medical research, biological research and related scientific research technical services, and professionally provide customized organoid culture services, offering standardized and traceable experimental support for research institutions and medical units.
Relying on our professional technical capabilities, we can customize organoid culture protocols according to customer needs, with strict whole-process control of experimental workflows to ensure the stability and reliability of culture results. We support the efficient development of related research such as genomics research, clinical trials and medical laboratory work, providing a strong impetus for scientific research innovation.








